Alluvi – Tirzepatide 40mg (R&D Only) | High-Purity UK Peptide
£110.00
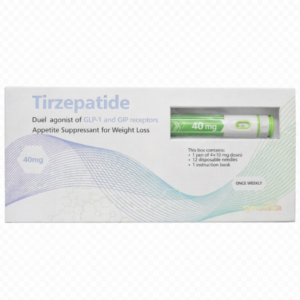
Alluvi – Tirzepatide 40mg Lyophilised Powder (R&D Use Only)
Premium Tirzepatide 40mg research peptide from the Alluvi series. Engineered for high-precision laboratory analysis and metabolic R&D projects. Each vial is lyophilised for maximum stability and supplied with a batch-specific Certificate of Analysis (COA).
-
- Purity: >99.2% (HPLC Verified)
- Format: 40mg Vacuum-Sealed Vial
- Origin: UK Laboratory Tested




Reviews
There are no reviews yet.